Selective
Targeting,
Thoughtful
Design
Scientific Overview
IN CANCER
Evolution of
cell therapy
in cancer
Century’s approach builds on the foundation of autologous and allogeneic cell therapy research to transform cell therapy as we know it today.
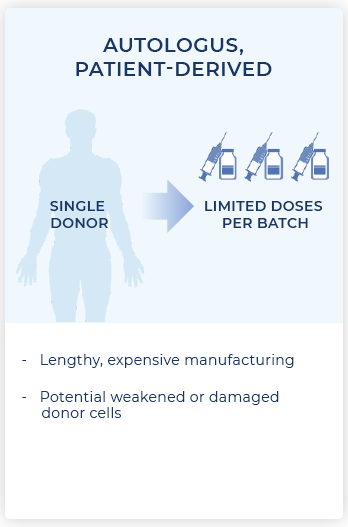
While autologous CAR-T therapies that create individual treatments from the cells of each patient have demonstrated success in treating certain blood cancers, there remain significant limitations to this approach. These include lengthy delays for treatment initiation due to manufacturing complexities and cost. Furthermore, the cells from which the product is made are often dysfunctional because they come from cancer patients.
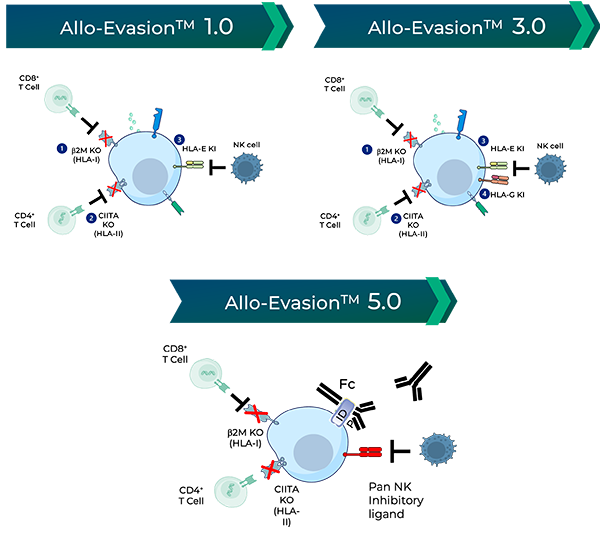
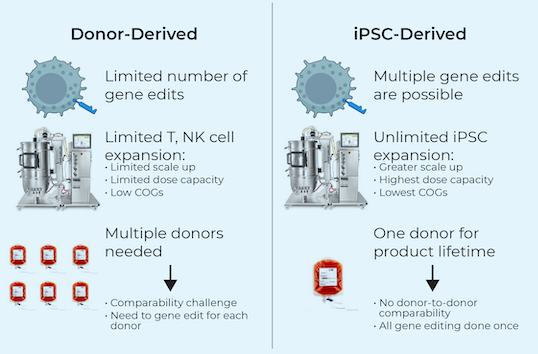
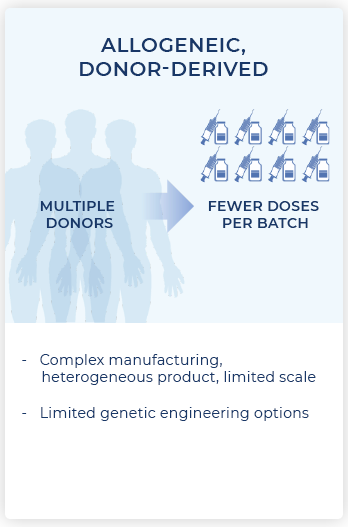
Allogeneic CAR-T therapies overcome some of these limitations by using healthy donor cells, but because the cells are foreign to the patient, they can be rejected by the patient’s immune system, or even attack normal cells in the patient. In addition, during manufacturing, these normal donor cells can only expand or “replicate” to a limited degree before becoming dysfunctional, requiring repeat collection of fresh cells from the donors to replenish drug inventory.
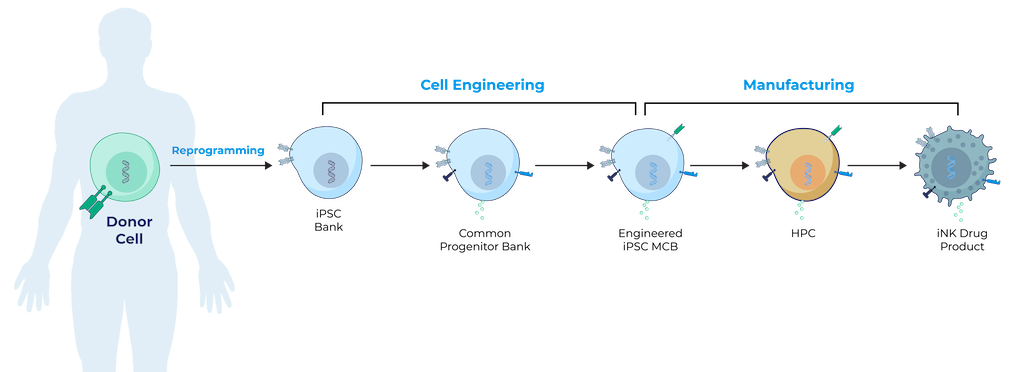
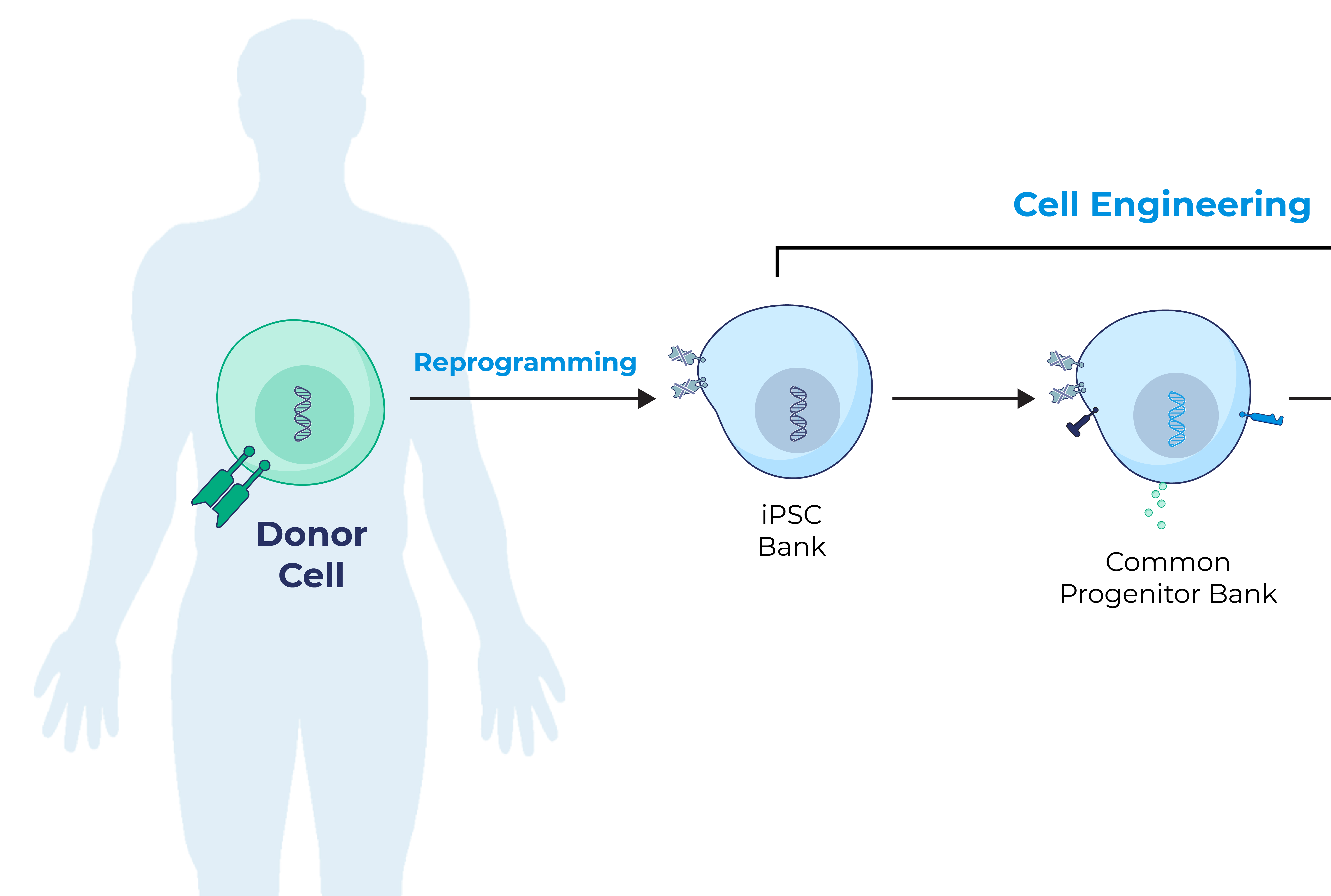
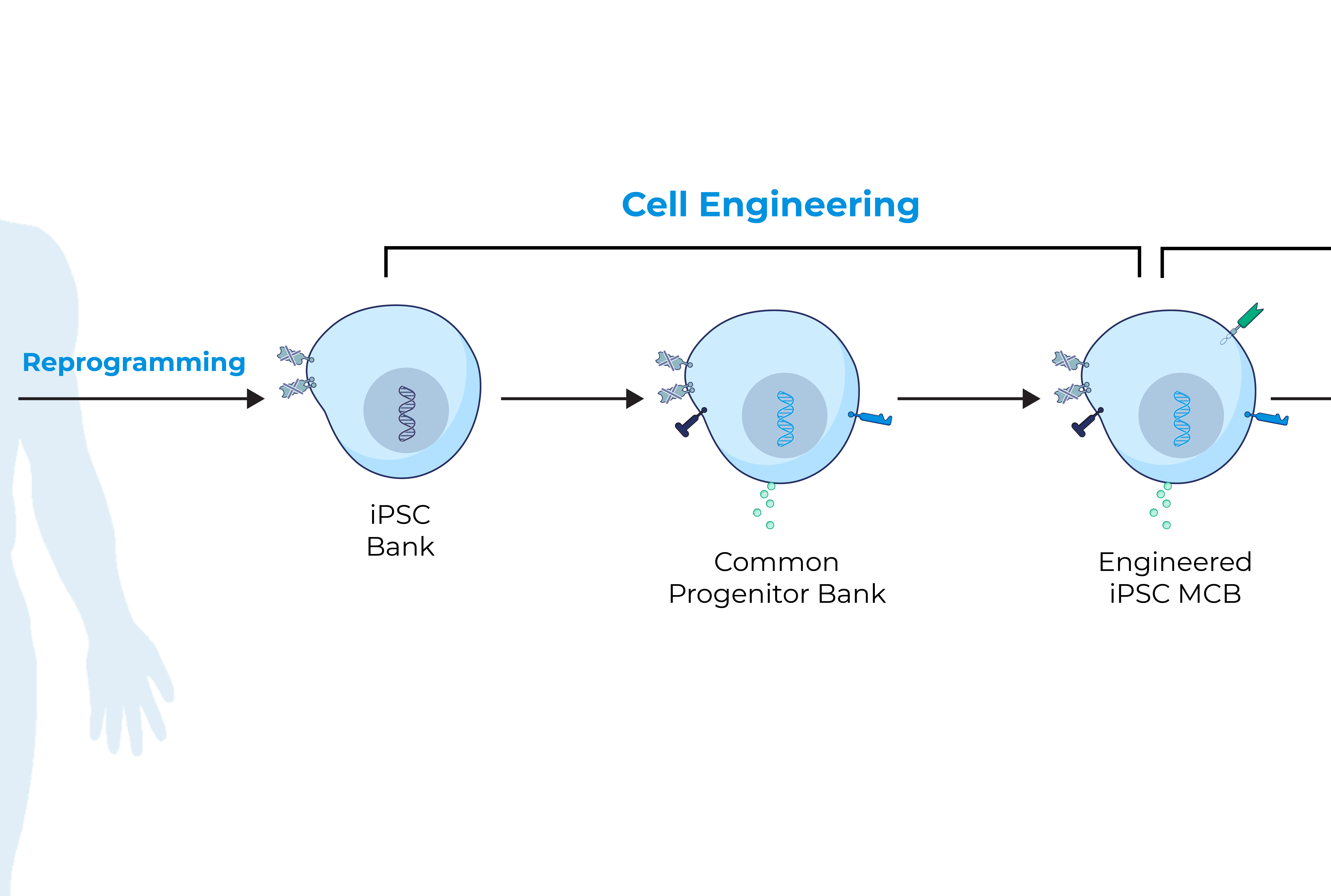
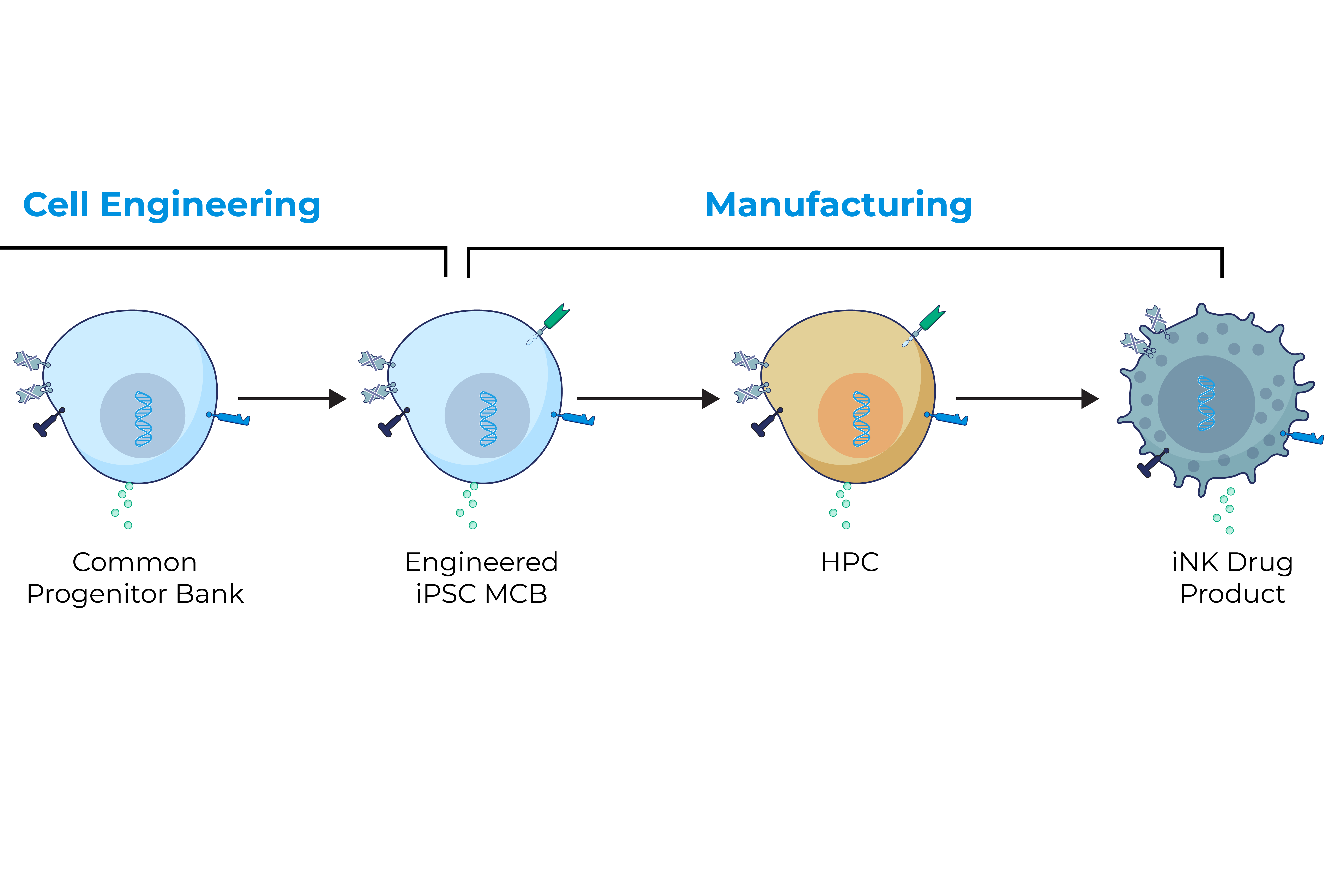
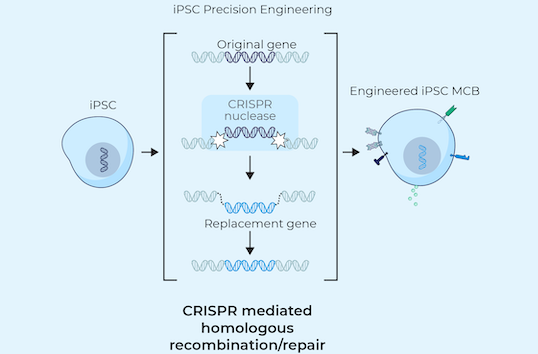
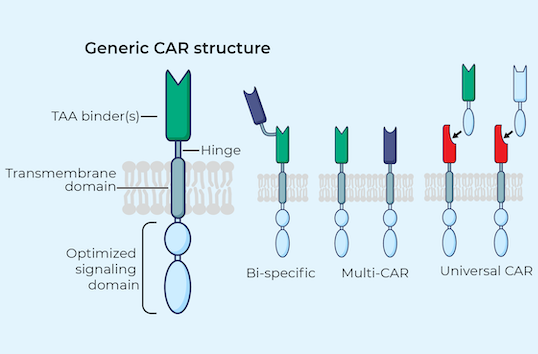
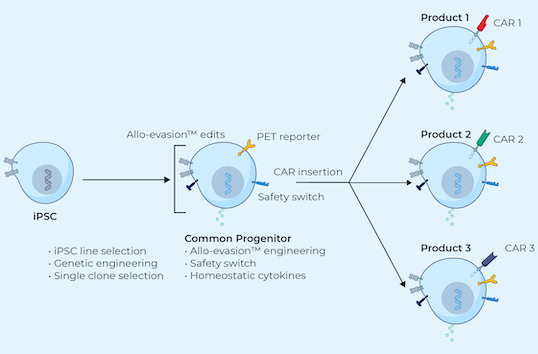
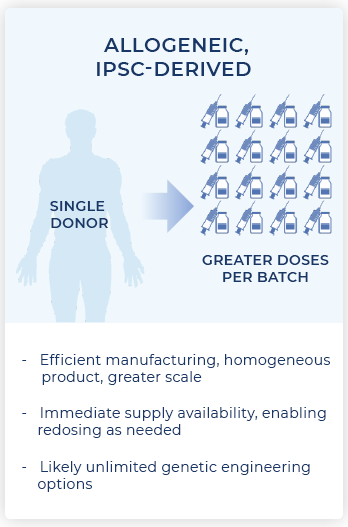
iPSCs provide donor cells with unlimited replication capacity, offering the potential for a more streamlined manufacturing process and consistent product. Century’s approach takes advantage of iPSCs engineered with common features, which act as a “platform” for generating an army of cancer-fighting NK and T cell products. This approach eliminates the need to repeat these engineering steps for future programs, saving time, maximizing efficiency, improving quality control, and enabling potentially rapid development of multiple cell products.


Century’s Approach